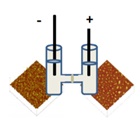
One-step cathodic and anodic synthesis of hydrophilic carbon nanomaterials
It was demonstrated that the galvanostatic polarization of graphite electrodes in a two separated compartments cell generates simultaneously and straightforwardly, without further separation and functionalization, two distinct sets of hydrophilic carbon nanomaterials (EHC – Electrogenerated Hydrophilic Carbon), in anodic and cathodic compartments, respectively. The synthesized products comprise an amorphous carbonaceous material with aromatic sp2 carbon clusters, differing mainly on their oxidation degree. Surprisingly, it wasfound that nitrogen is incorporated in the carbon-based material along the electrolysis, which is provided by the N2 gas present in the air and/or aqueous medium. This phenomenon is explained on basis that very reactive carbonaceous species are formed during the electrodes polarization due to an anomalous high electric field generated along the galvanostatic polarization. In addition, it was also evidenced that ECH materials display a negative charge counterbalanced by sodium ions and unique optical properties, exhibiting deep ultraviolet emission (∼4.1 eV), as well as blue luminescence. It is foreseen that these carbon materials will have important repercussions in the optoelectronics devices, energy technology (supercapacitors, batteries and fuel cells) and biomedicine.
https://doi.org/10.1002/celc.201700386
Autores: A. D. Veloso, A. M. Botelho do Rego, A. M. Ferraria, L. F. V. Ferreira, D. P. Ferreira, P.B. Tavares , R. Videira, A. S. Viana , M. Cristina Oliveira